David Schrum


Our department emphasizes hands-on learning and close interaction between students and faculty. From the first semester of general chemistry to the senior-level advanced laboratory course, students learn the theoretical concepts and laboratory skills important to being well-educated chemists. Students learn to read primary literature, plan experiments, acquire and analyze data, and gain direct experience with a variety of modern instruments. We also place a strong emphasis on scientific writing and oral presentation skills.
The chemistry department has offered an American Chemical Society (ACS) approved program since 1953 and is certified to meet the requirements for professional training in chemistry established by the Committee for Professional Training of the ACS.

All chemistry majors undertake an original, faculty-mentored research project by the May term of their junior year, culminating in a thesis and seminar presentation. Many students also participate in the Summer Science Research program, present at regional and national meetings, and publish in peer-reviewed journals.
Our chemistry students, from first year through senior year, have hands-on access to advanced research-grade instrumentation—access typically reserved for graduate students or faculty at other universities. See list →
Most classrooms are equipped with smart projection technology, and all teaching laboratories feature constant-volume fume hoods and deionized water. We also have environmental rooms, darkroom facilities, a dedicated computing lab with molecular modeling software, a centralized instrument lab, and a laser laboratory.
Our dedicated professors bring extensive expertise to the field, earning awards, publishing impactful research, and sharing their knowledge as invited speakers. Most importantly, they are invested in mentoring students, supporting their growth, and guiding their academic journey.

Discover theoretical concepts, develop skills in data analytics and experimentation processes, or explore the intersection of biology and chemistry.

Both minors are designed to provide you with basic expertise in chemistry, while the environmental chemistry minor additionally covers environmental topics.

College of Arts and Sciences (CAS) undergraduate student interested in a future career as a Chemistry teacher.























— Andrea Carrillo ’25, B.S. in Chemistry

Shaun Weatherly '22, who is working toward a degree in biochemistry, spent his summer working with Dr. Rebecca Lyons.


Every year, the University of Redlands hosts the Student Science Research Program, where about 30 students take part in hands-on, immersive research alongside faculty mentors. During the 10-week summer program, students live on campus and receive a stipend for their work, made possible through endowment support from the John Stauffer Charitable Trust and other generous donors. Learn more about what a few students have to say about their experience.
A collection of research projects presented by the students and faculty from the Department of Chemistry.

Instrumentation list
See all instruments available for teaching labs and research.
The mission of the ACS Chemistry Club is to spread environmental awareness and chemical knowledge within the Redlands community. The club welcomes individuals of all identities and endeavors to foster a sense of community within and outside the realm of science.
* = University of Redlands undergraduates are coauthors.
Dr. David Brook (San Jose State University), David Soulsby (University of Redlands), CONTRAST AGENTS FOR ENHANCED MAGNETIC RESONANCE IMAGING. PCT/ US 2025/ 048729 - 63/700, 901, 2024.
Glover, R.; Soulsby, D. "One-pot Dess-Martin periodinane-mediated oxidative deprotection and olefination of trimethylsilyl-protected pyranosides and pyranoses", Carb. Res. 2023, 523, 108904.
T. Longin “Using the HSQC Experiment to Teach 2D NMR Spectroscopy in Physical Chemistry”, In NMR Spectroscopy in the Undergraduate Curriculum, Vol. 4; Soulsby, D,; Anna, L. J.; Wallner, A. S., Eds.; ACS Symposium Series; American Chemical Society; Washington, D. C., 2022; pp 175-189.
NMR Spectroscopy in the Undergraduate Curriculum: In-Person and Distance Learning Approaches, Volume 4; Soulsby, D.; Anna, L., J.; Wallner, A. S., Eds.; ACS Symposium Series 1376; American Chemical Society: Washington, DC, 2021. Distributed by Oxford University Press.
Soulsby, D. "Using 1H NMR Spectroscopy to Study the Free Radical Chlorination of Alkanes" J. Chem. Educ., 2020, 97, 2286-2290.
Soulsby, D. Band‐selective excitation NMR spectroscopy and quantitative time‐domain analysis using Complete Reduction to Amplitude‐Frequency Table (CRAFT) to determine distribution coefficients during drug development, Magn. Reson. Chem. 2019, 57(11), 953-960
Aziz, T.; Sardana, A.; Cottrell, B.; Soulsby, D. Dissolved Organic Matter Processing and Photoreactivity in a Wastewater Treatment Constructed Wetland, Science of the Total Environment, 2019, 648, 923-934,
A.C. Vander Wall, P. Lakey, E. Rossich-Molina, V. Perraud, L. M. Wingen, J. Xu, D. Soulsby, R. B. Gerber, M. Shiraiwa, and B. J. Finlayson-Pitts, Understanding Interactions of Gases with the Surface and Bulk of Organic Films: Implications for Particle Growth in the Atmosphere published in themed collection: Atmospheric Surfaces - Environmental Science: Processes and Impacts, 2018, 20, 1593-1610
S. Kumbhani, T. Longin, L. M. Wingen, C. Kidd, V. Perraud, B. J. Finlayson-Pitts, “New Mechanism of Extractive Electrospray Ionization Mass Spectrometry for Heterogeneous Solid Particles”, Analytical Chemistry, 2018, 90, 2055.
S. Malaker, M. Ferracane, F. Depontieu, A. Zarling, J. Shabanowitz, D. Bai, V. Engelhard, S. Topalian, D. Hunt, D. “Identification and characterization of complex glycosylated peptides presented by the MHC class II processing pathway in melanoma.” Journal of Proteome Research 2017, 16, 228-237.
*David Soulsby, Jeryl Anne Chica "Determination of Partition Coefficients Using 1H NMR Spectroscopy and Time Domain Complete Reduction to Amplitude-Frequency Table (CRAFT) Analysis" Magnetic Resonance in Chemistry, 2017, 55, 724-729.
Rebecca A. Lyons, Lisa Benevenuti, “Deposition and distribution factors for the endocrine disruptor, 4-nonylphenol, in the Sierra Nevada Mountains, California, USA”, Journal of Environmental and Analytical Toxicology, 2016, 6, 388, doi:10.4172/2161-0525.1000388.
*Rebecca A. Lyons, Blodwyn McIntrye, Lissah Jensen, “Phosphorus loading rates in lakes with development and stocked fish in the Sierra Nevada Mountains, California, USA”, Ecosphere, 2016, 11, doi: 10.1002/ecs2.1554.
David Soulsby, “Application of 1H and 1D TOCSY NMR Spectroscopy to the Free Radical Chlorination of Alkanes Experiment” in NMR Spectroscopy in the Undergraduate Curriculum: First Year and Organic Chemistry Courses, Soulsby, D.; Anna, L., J.; Wallner, A. S., Eds.; ACS Symposium Series 1221; American Chemical Society: Washington, DC, 2016
Anthony Wallner, David Soulsby, “Introduction to NMR Spectroscopy in the Undergraduate Curriculum”, in NMR Spectroscopy in the Undergraduate Curriculum: First Year and Organic Chemistry Courses, Soulsby, D.; Anna, L., J.; Wallner, A. S., Eds.; ACS Symposium Series 1221; American Chemical Society: Washington, DC, 2016.
*Rebecca Lyons, Steven Moore, David Smith, and Kyle Van de Bittner, "Mile-High Chemistry: Spatially Enabled Water Quality Research in California." In STEM and GIS in Higher Education, edited by David Cowen. Redlands, California: Esri Press, 2016.
*Rebecca Lyons, Kyle Van de Bittner, Sean Morgan-Jones, “Deposition patterns and transport mechanisms for the endocrine disruptor 4-nonylphenol across the Sierra Nevada Mountains, California”, Environmental Pollution, 2014, 195,123-132.
David Soulsby, “Modern NMR Experiments: Applications in the Undergraduate Curriculum”, in NMR Spectroscopy in the Undergraduate Curriculum, Soulsby, D.; Anna, L., J.; Wallner, A. S., Eds.; ACS Symposium Series 1128; American Chemical Society: Washington, DC, 2013.
Anthony Wallner, Laura Anna, David Soulsby, “Introduction to NMR Spectroscopy in the Undergraduate Curriculum”, in NMR Spectroscopy in the Undergraduate Curriculum, Soulsby, D.; Anna, L., J.; Wallner, A. S., Eds.; ACS Symposium Series 1128; American Chemical Society: Washington, DC, 2013.
Rebecca A. Lyons, John P. Hassett, Anna M. Flach, Israel Cabasso, " Development of Thin Layer Polymers to Concentrate and Detect Aquatic Contaminants", Journal of Applied Polymer Science, 2012, DOI: 10.1002/app.37857.
David Soulsby, "Using Cloud Storage for NMR Data Distribution", Journal of Chemical Education, 2012, 89, 1007-1011.
Lambert A. Doezema, Teresa Longin, William Cody, Veronique Perraud, Matthew Dawson, Michael J. Ezell, John Greaves, Kathleen R. Johnson, Barbara J. Finlayson-Pitts, " Analysis of secondary organic aerosols in air using extractive electrospray ionization mass spectrometry (EESI-MS)", RSC Advances, 2012, 2, 2930-2938.
Ryan A. Picou, David P. Schrum, Ginger Ku, Rebekah A. Cerqua, Indu Kheterpal, S. Douglas Gilman, " Separation and detection of individual Ab aggregates by capillary electrophoresis with laser-induced fluorescence detection", Analytical Biochemistry, 2012, 425, 104-112.
*Teresa Longin, John Terhorst, Chris Lang, "Simulations of Photopumping in Doubly Illuminated Liquid Membranes Containing Photoactive Carriers", Journal of Physical Chemistry B, 2010, 114, 15846.
J. R. Blauth, D. P. Schrum, "Service Learning and Field Research at the University of Redlands: Desert Restoration in Joshua Tree National Park", CUR Quarterly, 2005, 26, 63-65.
*Teresa L. Longin, Teresa Fraterman, Phuong Nguyen, "Simulations of photomodulated solute transport in doubly illuminated liquid membranes containing photoactive carriers", Journal of Physical Chemistry B, 2005, 109, 21063-21077.
Michelle L. Goyette, Teresa L. Longin, Richard D. Noble, Carl A. Koval, " Selective photofacilitated transport of sodium ions through liquid membranes: key factors in experimental design, transport results and comparison with a mathematical model", Journal of Membrane Science, 2003, 212, 225-235.
Books
NMR Spectroscopy in the Undergraduate Curriculum: Upper-Level Courses and Across the Curriculum, Volume 3; Soulsby, D.; Anna, L., J.; Wallner, A. S., Eds.; ACS Symposium Series 1225; American Chemical Society: Washington, DC, 2016 Distributed by Oxford University Press.
NMR Spectroscopy in the Undergraduate Curriculum: First Year and Organic Chemistry Courses, Volume 2; Soulsby, D.; Anna, L., J.; Wallner, A. S., Eds.; ACS Symposium Series 1221; American Chemical Society: Washington, DC, 2016 Distributed by Oxford University Press.
NMR Spectroscopy in the Undergraduate Curriculum, Volume 1; Soulsby, D.; Anna, L., J.; Wallner, A. S., Eds.; ACS Symposium Series 1128; American Chemical Society: Washington, DC, 2013 Distributed by Oxford University Press.
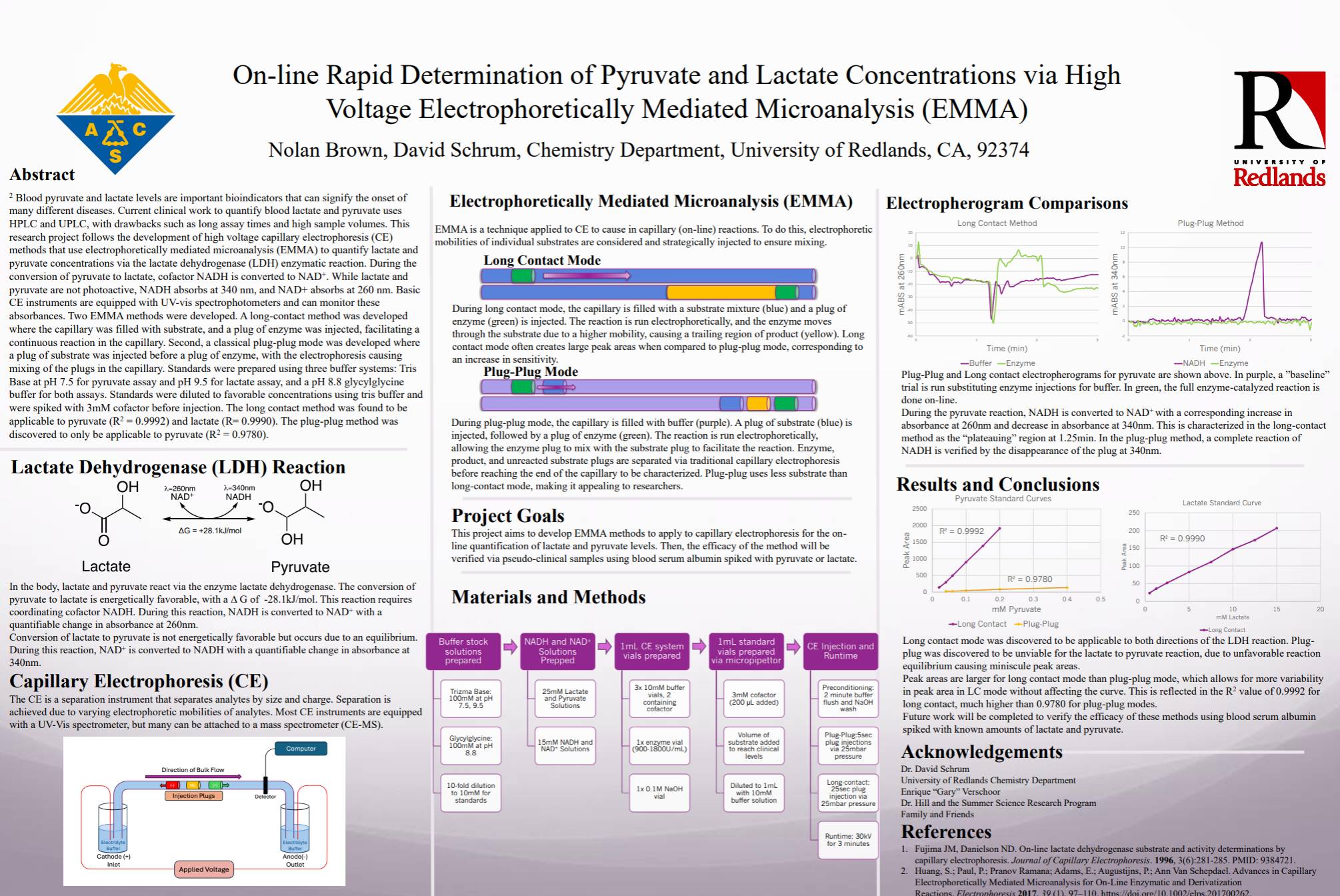
N. Brown, D. P. Schrum, “Online Analytical Determination of Pyruvate and Lactate Concentrations via Electrophoretically Mediated Micro-Analysis (EMMA),” presented at the American Chemical Society National Meeting, San Diego, CA, March 2025.
A. Klausner, R. Lyons, D.P. Schrum, “An Analytical Determination of Kidney and Bladder Stone Causing Minerals and Compounds in Dog Foods,” presented at the Southern California Conference on Undergraduate Research (SCCUR), California State University – Fullerton, November 2023.
L. Ionescu, J.L. Krstenanansky, D.P. Schrum, “The Isolation and Characterization of the Four Major Alkaloids in Sceletium tortuosum, an Alternative Natural Therapeutic for Anxiety and Depression”, presented at the American Chemical Society National Meeting, San Diego, CA, March 2022.
*Glover, R.; Soulsby, D. "Trimethylsilyl-protected pyranosides and pyranoses: New approaches to regioselective olefination and etherification reactions," poster presentation, at the American Chemical Society National Meeting and Exposition.
*Huynh, T.; Soulsby, D.; Longin, T. "Characterization of trace compounds in craft and commercial gins using liquid chromatograph-mass spectrometry," poster presentation, at the American Chemical Society National Meeting and Exposition.
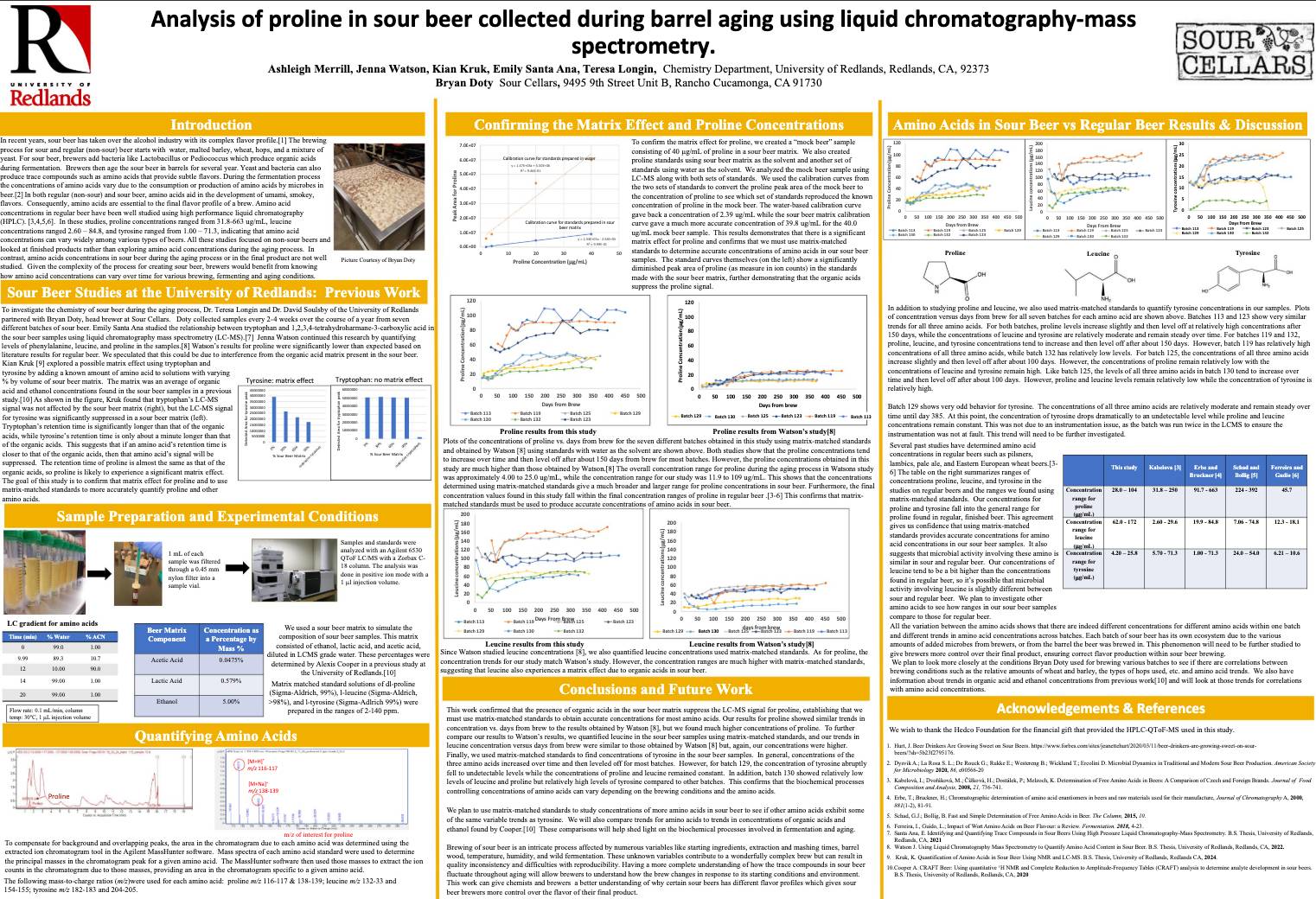
*Higgins, K.; Watson, J.; Doty, B.; Soulsby, D.; Longin, T. "Analysis of hordatines and amino acids in sour beer using liquid chromatography-mass spectrometry poster presentation", at the American Chemical Society National Meeting and Exposition.
*Silveira, L. A.; Hoxha, D.; Dickson, O.; Opara, S.; Dialynas, A.; Olsson, S.; Soulsby, D.; Liao, J.; Shih, J.; Curtin, N.; Venable, M. "Identification of genes influencing production of pulcherrimin, an iron-binding pigment, in the yeast Kluyveromyces lactis", at Cell Bio Virtual 2020.
*Longin, T.; Soulsby, D.; Cooper, A.; Santa Ana, E., Doty, B. "Analysis of organic acids and trace compounds in sour beer using NMR spectroscopy and LC/Q-Tof MS", poster presentation at the 260th American Chemical Society Virtual Meeting and Exposition.
*Soulsby, D.; Doty, B.; Cooper, A. "CRAFT sour beer: Quantification of metabolites over time in spontaneously fermented beer using quantitative 1H NMR spectroscopy and time-domain analysis", poster presentation at the 259th National American Chemical Society Meeting and Exposition, Philadelphia, PA.
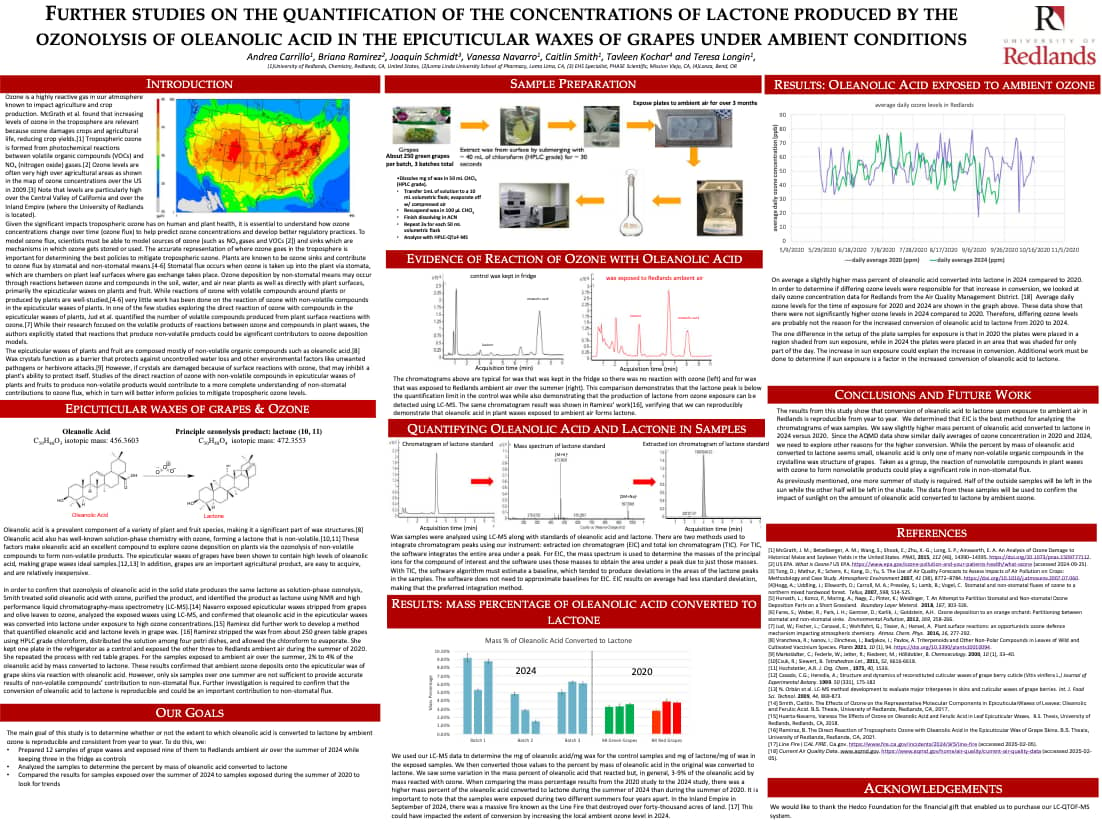
*Longin, T. L.; Huerta Navarro, V.; Romero, B.; Smith, C.; Rogoff, T.; Kochar, T.; Soulsby, David P. "Evidence of direct reaction between ozone and oleanolic acid in plant cuticular waxes under laboratory and ambient conditions", poster presentation at the 258th National American Chemical Society Meeting and Exposition, San Diego, CA.
Soulsby, D. "Sharing NMR data in the cloud: Expanding the opportunities for learning in the undergraduate laboratory", oral presentation at the 257th National American Chemical Society Meeting and Exposition, Orlando, FL.
*Hughes, C.; Stubb, N.; Soulsby, D.; Wacks, D. "Determining the Carbon Source Hierarchy of Sinorhizobium meliloti using 1H NMR Spectroscopy and CRAFT", poster presentation at the 255th National American Chemical Society Meeting, New Orleans, LA.
*C. Hughes (presenter), N. Stubb, D. Soulsby, D. Wacks, “Determining the carbon source hierarchy of Sinorhizobium meliloti using 1H NMR Spectroscopy” poster presentation at the Southern California Conferences for Undergraduate Research, California State Polytechnic University, Pomona, CA, November 18, 2017.
*N. Stubb (presenter), D. B. Wacks, D. Soulsby, “Quantitating Metabolite Consumption by Sinorhizobium meliloti Using 1H NMR Spectroscopy”, poster presentation at the 253rd National Meeting of the American Chemical Society, San Francisco, CA, April 2017.
*C. Smith (presenter), M. J. Riches, T. Kochar, D. Soulsby, T. Longin, “The effects of ozone on representative molecular components in epicuticular waxes of leaves: Oleanolic and ferulic acid”, poster presentation at the 253rd National American Chemical Society Meeting, San Francisco, CA, April 2017, Section: Analytical.
T. Longin, “Using a guided inquiry and experimental approach to teaching vibrational-rotational spectroscopy”, poster presentation at the 253rd National American Chemical Society Meeting, San Francisco, CA, April 2017, Section: Chemical Education.
*S. Finkbohner (presenter), D.P. Schrum, “Electrophoretically Mediated Microanalysis (EMMA): an Application of Capillary Electrophoresis for Performing Enzyme Assays”, presented at the Southern California Conference on Undergraduate Research (SCCUR), University of California-Riverside, November 2016.
*D. Santeliz, J.L. Krstenanansky, D.P. Schrum (presenter), “Analysis and Characterization of Mesembrine-type Alkaloids obtained from Sceletium tortuosum”, poster presentation at the 251st American Chemical Society National Meeting, San Diego, CA, March 2016.
D. Soulsby, “CRAFT Beer: Quantitation of the Major Components of Commercially Available Sour Beers Using Time-Domain CRAFT Analysis of 1H NMR Spectra”, poster presentation at the 251st National Meeting of the American Chemical Society, San Diego, CA, March 2016.
*Lissah Jensen (presenter), Rebecca Lyons, Wendy McIntyre, “Phosphorus loading rates in lakes with shoreline development and stocked fish in the Sierra Nevada Mountains, California, USA”, poster presentation at the American Geophysical Union National Meeting, San Francisco, CA, December 2015.
*Andrew Schlaus (presenter), Rebecca Lyons, “Solid Phase Microextraction with Polystyrene- Poly(dimethylsiloxane) block copolymers”, presentation at the 249th American Chemical Society National Meeting, Denver, CO, March 2015.
Teresa L. Longin, "Development of a guided-inquiry laboratory activity to introduce major concepts in chemical equilibrium for general chemistry", oral presentation at the 243rd National Meeting of the American Chemical Society, San Diego, CA, March 2012.
Rebecca Lyons, "Developing a predictive model for gasoline and gasoline by-products in surface waters of Southern California", oral presentation at the 243rd National Meeting of the American Chemical Society, San Diego, CA, March 2012.
David Soulsby, "Using Experimental and Computational Chemistry to Teach Electrophilic Aromatic Substitution Reactions", poster presentation at the 243rd National Meeting of the American Chemical Society, San Diego, CA, March 2012.
Teresa Longin, Lambert Doezema, William Cody, Veronique Perraud, Michael Ezell, Matthew Dawson, John Greaves, Barbara J. Finlayson-Pitts, "A New Approach to Speciation of the Organic Component of Atmospheric Aerosols", poster presentation at the American Geophysical Union Fall Meeting, San Francisco, December 2011.
Rebecca Lyons, John P. Hassett, "A polymer-based thin film trap and optical detection system for hydrophobic compounds in water", oral presentation at the PacifiChem Conference, Honolulu, HI, 2010.
*David Soulsby, Cesar B. Rodarte, "Substituent effects on the E/Z alkene ratio in medium-membered rings synthesized via RCM", poster presentation at the 239th National Meeting of the American Chemical Society, San Francisco, CA, March 2010.
*L. Hensleigh, David Schrum, "Characterization of Soil from Joshua Tree National Park", 18th Annual Southern California Conference on Undergraduate Research, Pepperdine University, November 2010.
*Matt Steele, David Schrum, "Analysis of ß-Amyloid Peptide Fragments Involved in Alzheimer's Disease via Capillary Electrophoresis (CE) and High-Pressure Liquid Chromatography-Mass Spectrometry (HPLC-MS), 18th Annual Southern California Conference on Undergraduate Research, Pepperdine University, November 2010.
Our faculty combine academic expertise with real-world experience, mentoring students through coursework, research, and career development.